DOW CHEMICALS
Dow Chemicals – Manufacturing Toxins and Breast Implants
What is it about powerful US companies and their tendency to do absolutely anything to maximise their profits with no regard to how many people they kill off if it makes them a buck. Could it be that they are just like the American government? Could it be that they actually run the American government?…. It certainly looks like they are helping to fund it….. Without doubt, Dow blows goats, and silicone breast implants are not a good idea, even if they did work wonders for Pamela Anderson…

DOW CHEMICALS
Number two in chemical sales in the U.S. Employees: 58,000. Sales: $20 billion. Headquarters: Midland, Michigan, U.S.

Dow, the manufacturer of Napalm and Agent Orange during Vietnam War, and now the target of a billion dollars worth of lawsuits over their highly destructive silicone breast implants, is partners with the drug firm Ely Lilly in Dow Elanco, a spinoff company that is the largest producer of insecticides and fungicides in the U.S.
Dow must have a magnetic attraction for severe defoliants. Having distanced itself from Agent Orange — its partner Lilly now makes Tebuthiuron, an herbicide that kills soil so that no plants can grow on it in the future. Sounds like a weapon of war.
Of course Dow also tries to distance itself from dioxin (contained in tis Vietnam era Agent Orange), but Greenpeace reports that hugely produced chlorine based Dow products — pesticides, solvents and PVC plastics — are the single largest source in the world of dioxin today.
Dow owns Marion Merrell Dow (MMD), a major pharmaceutical house. Like all drug companies, whether you know it or not, the commercial output of MMD is chillingly toxic. Let’s start there.
Examples:
-
MMD’s vaginal supository AVC cream is used to treat candida albicans. The PDR states that there is no data available on the long term potential of AVR for causing cancer or birth defects, but “deaths associated with administration of oral sulfonamides (such as AVC) have reportedly occured form hypersensitivity reactions, agranulocytosis, aplastic anemia and other blood discrasias.” . . . Comforting.
-
Bentyl, Dow’s drug for irritable bowel syndrome, also has in the PDR listing “no known data” for long term potential carcinogenicity or birth defects, but “psychosis has been reported in sensitive individuals.” There are also, the PDR says, reports of deaths from respiratory collapse.
-
Cardizem, the Dow drug for hypetension and angina, carries the PDR caution: “Worsening of congestive heart failure has been reported in patients with preexisting impairment of ventricular function.”
Nothing could prepare a sane person for the PDR’s description of Dow’s Clomid, a drug that attempts to produce ovulatory stimulation so that pregnancy can occur in women for whom that would otherwise be unlikely.
Here is a partial list of Clomid’s post-marketing adverse effects:
- seizure
- stroke
- psychosis
- cataracts
- posterior vitreous detachment
- arrhythmia
- tachycardia
- hepatitis
- liver, breast, pituitary, ovarian, kidney, tongue & bladder cancer
- brain abscess
- tubal pregnancy
- uterine hemorrhage
- ovarian hemorrhage
In the babies born to the mothers taking Clomid, there have occurred:
- neuroectodermal tumor
- thyroid tumor
- leukemia
- abnormal bone development including skeletal malformations of the skull, face, nasal passages, jaw, hand, limb and foot joints
- malformations of the anus, eye, lens, ear, lung, heart and genitalia
- dwarfism
- deafness
- mental retardation
- chromosomal disorders
- neural tube defects
Lorelco, Dow’s drug aimed at lowering cholesterol, has this ominous PDR caution: females should be warned not to become pregnant for at least six months after discontinuing Lorelco. Lorelco’s adverse effects?
- Gastrointestinal bleeding
- vomiting
- low hemoglobin
- fetid sweat
- impotency
- anorexia
- diminished sense of taste and smell.
Dow makes Norpramin, an antidepressant. The PDR states: “It is important that this drug be dispensed in the least possible quantities to depressed outpatients since suicide has been accomplished with this class of drug.”
Some of the effects of Norpramin are:
- both elevating and lowering of blood sugar levels
- heart block, myocardial infarction, stroke
- sudden death
- hallucinations, delusions
- tremors, ataxia, peripheral neuropathy, seizures
- dilation of urinary tract
- bone marrow depression
- vomiting, black tongue, hepatitis
- impotence, painful ejaculation, testicular swelling
- weight gain or loss.
(Note: In these drug summaries I don’t even bother to comment about the uniform unworkability of the drugs on the causes of the illnesses for which they are prescribed nor will I comment on a further danger: the effects of combining several drugs at once. Nor on the fact that OTHER non-toxic remedies and approaches to health would eliminate the need for these drugs and their poisonous effects.)
Dow makes Rifadin, a “semi-synthetic” antibiotic for the treatment of tuberculosis. The PDR comments, “Rifadin has been shown to produce liver dysfunction. Fatalities associated with jaundice have occurred in patients with (previous) liver disease.” The PDR further issues a bizarre warning — “Rifadin can cause the urine, feces, saliva, sputum, sweat, and tears to turn red-orange. “Permanent discoloration of soft contact lenses may occur.”
The suggested Rifadin dosage for people with TB is 600mg a day for six to nine months. Yet the PDR gives this warning: “High doses of Rifadin greater than 600mg given once or twice a week have resulted in high incidence of adverse reactions, including leukopenia (abnormal decrease in white blood corpuscles), thrombocytopenia (abnormal decrease in blood platelets), acute hemolytic anemia, shock, renal failure.”
Among Rifadin’s other advese effects are anorexia, vomiting and menstrual disturbances.
I have tried in listing adverse effects to avoid dipping into the explicit PDR category “rare” and the category, “has been found to occur in less that 1% of people taking drug and vanishes upon discontinuing drug.” That leaves the open categories of “general adverse effects” or “we don’t really know how many people on the drug suffer from these effects” or the “these effects are reported to occur after drug is marketed to the public and there is no way to prove the effects are caused by the drug.” I have relied for the most part on these three last categories.
Dow and Ely Lilly and Company of Indianapolis are partners in a corporation called Dow Elanco, one of the largest producers of agricultural chemicals in the world. As a 40 percent partner Lilly falls within the purview of Dow and so I have justifiably included its drug products under the umbrella of Dow in this section.
- Lilly manufactures Heparin sodium (derived from the intestinal mucosa of pigs), a blood anticoagulant used to prevent clotting. Says the PDR, “hemorrhage can occur at virtually any site in patients receiving Heparin. Patients on the drug can develop an “irreversible aggregation of (blood) platelets . . . (which) may lead to gangrene of the extremities . . . (and) amputation, mycardial infarction, pulmonary emoblism, stroke and possibly death.”
- Lilly’s Nalfon is an NSAID for (non-steroidal anti-inflammatory drug). Every year in the U.S. seven to eight thousand people die from the administration of NSAIDs and between 70,000 to 80,000 are hospitalized from their use.
- Lilly’s Prozac is the wildly popular “in” anti-depressant of the moment. Prior to its release, it was never tested on humans for longer than thirteen weeks. Prozac has been associated with suicidal and murderous behavior, and the dampening of sexual desire. Its other effects include insomnia, anxiety, and anorexia (in 9 percent of the patients in clinical trials). Fifteen percent of the 4,000 patients who received Prozac in pre-release clinical trials discontinued treatment due to “an adverse event.”
- Diethylstilbestrol, a Lilly drug, is a synthetic estrogenic substance used for breast cancer and prostate cancer (as a palliative only). The PDR states, “WARNING: USE OF ESTROGENS HAS BEEN REPORTED TO INCREASE THE RISK OF ENDOMETRIAL CARCINOMA. ESTROGENS SHOULD NOT BE USED DURING PREGNANCY. ITS USE MAY CAUSE SEVERE HARM TO THE FETUS. More PDR quotes on this drug:
- “A recent study reported a two to threefold increase in the risk of gall bladder disease occuring in women receiving post-menstrual estrogen therapy . . . “
- “In a large prospective clinical trial in men, large doses of estrogen . . . comparable to those used to treat cancer of the prostrate . . . have been shown to increase the risk of non-fatal myocardial infarction, pulmonary emobilism. . . ”
Adverse reactions to diethylstilbestrol include breakthrough bleeding, spotting and change in menstrual flow; vomiting; cholestatic jaundice; hemorrhagic skin eruption; corneal curvature; and migraine.
All these effects for a cancer treatment that is admittedly only a palliative?
(Note: The January 28, 1994, Congressional Quarterly in its report, Regulating Pesticides, points out that pollutants in the environment are being found to contain estrogenic substances. And that several researchers have linked exposure to estrogens with cancer, including breast cancer. (Now read the above section the drug diethlystilbestrol again and if you mind isn’t completely blown, check your breath on a mirror.)
The above list and description of medical drugs is certainly not meant to be all-inclusive vis-a-vis Dow. It is just a bitter sample. If you find yourself saying, “Well, even if these drugs have some horrible effects, the doctors who prescribe them must know what they’re doing”, consider that once people said exactly that about the U.S. corporations who were busy spilling poisonous chemicals into the rivers of this land. “They must know what they’re doing. They would never . . .” But they did. And these corporations are manufacturing the kinds of medical drugs I’ve just been describing AND the industrial chemicals AND the pesticides. Wake up and smell the poisons!

Half a billion pounds of toxic waste per year
Who could present a complete and specific portrait of Dow’s yearly industrial wastes? Inform, Inc. (New York City) has done an analysis of quantity in its Toxics Watch 1995 report. It culls the top twenty corporations from a total of 10,840 parent companies in the U.S. Dow ranks sixth in “production-related toxic chemical wastes, carcinogens and ozone depleting chemicals . . .”. How many pounds of waste are we talking about defecated by Dow into the world? 517.5 million pounds for 1992! Half a billion pounds.
Susan Cooper of the National Coalition Against the Misuse of Pesticides names Dow’s pesticide Dursban as a serious creator of health problems: nausea, headaches, behavioral changes in children. She told the Multinational Monitor that at least one out of every two phone calls that her office takes about pesticide complaints concerns Dursban. The Pesticide Action Network states that Dow produced or sold three pesticides on their “Dirty Dozen” list before 1980. One of these DBCP, ordered to be phased out by the EPA, now shows up being sold by Dow to the Dole Corporation, which has used it on its banana plantations in Costa Rica. DBCP contaminated ground water for several thousand square miles in the California central valley and caused sterility in agricultural workers. Four other Dow agricultural chemicals, Gallant, Verdict, Gauntlet, and Tridal, banned by the EPA, have shown up in Africa, Latin America, Central America, Asia and Europe.
Beyond the products mentioned so far, what to boycott made by Dow?
- Styrofoam labeled plastic products, agricutlural herbicides (Starane, Spike, Treflan), the soil fumigant Telone, and two insecticides, Dursban and Lorsban.
- It makes over-the-counter drugs: Norhistamine (cough), Cepacol, Gly-Oxide (antiseptic), Cepastat lozenges, Citrucel laxative, Delbrox (ear care), Gaviscon (antacid), the calcium supplement Os-Cal.
- Household products include Ziploc Bags, Fantastik Cleaner, Handi-Wrap, Saran Wrap, Spray ‘N Wash, Dow Bathroom Cleaner, Glass Plus Multi-Surface Cleaner, Smart Scrub, Ultra Yes laundry detergent, Vivid bleach and Style and Perma Soft hair products.
It should also be noted that Dow manufactures benzene, widely acknowledged as a carcinogen.
Of course, all this information is faxed and internetted around the world, people outside the U.S. will find the Dow subsidiaries in their countries and the products they make. In the U.S. the reference text The Directory of Multinationals is a good source for the names of these subsidiary corporations.
So to this point, you have much more than sufficient evidence of massive toxicity to justify a boycott of Dow. You can also see that boycotting their products is in some cases awkward, because wholesalers and companies, not individuals, are Dow’s customers. More reason to press disinvestment, making it unconscionable to hold stock of this company.

Did someone say “admitting liability for Bhopal”?

But don’t worry, breast implants are safe, healthy, and look great!
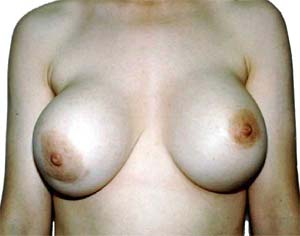
Here in New Zealand where American businesses take special care to look after our interests, there is compensation in the unlikely event that anything goes wrong with any silicon implants.
Kiwis paid out for bad implants
By RUTH HILL
A New Zealand breast cancer survivor, who suffered years of pain from a ruptured silicone implant, has finally received her payout from a multibillion-dollar compensation fund – a paltry $600.
The woman, who did not want to be named, said she was “surprised to get anything at all”, after a decade of legal wrangling. “Frankly, I had stopped hoping and just put it behind me.”
In 1999, a United States judge approved a $US6.4 billion ($NZ9.3 billion) settlement deal for worldwide compensation claims (including 1200 New Zealanders) against the American manufacturer Dow Corning. But overseas claimants, including the New Zealanders, were offered only 60% of the amount offered to Americans because they were unable to sue in the US. They appealed, but lost the case in 2002.
Now 564 claims by New Zealand women of between $NZ872 and $17,442 have been settled. It is not known how many claims are still outstanding because some individuals had more than one claim.
In November, the United States Food and Drug Administration (FDA) allowed silicone breast implants to be sold again, 13 years after they were removed from the market because of safety concerns.
While silicone breast implants have remained available in New Zealand, the Ministry of Health does not endorse their use and women must be warned of the risks before consenting to the operation.
The New Zealand woman was persuaded by her doctor to have silicone implants after a double mastectomy in 1984. She was told they would last “forever”.
But within seven years, one implant had ruptured, causing toxic silicone to seep into the surrounding tissue, disfiguring the breast and slowly poisoning her.
“Where the silicone was leaking, hard tissue formed around it, so I had lumps under my arms and all over the place,” she said.
She suffered crippling fatigue, insomnia, night sweats, colitis, numbness in her hands and feet, and her hair started falling out.
Yet it was three years before she could persuade her GP or the surgeon that anything was wrong. Her implants were removed in 1994 – 10 years after they were inserted.
She was unable to work. She could not lift her arms over her head because of the pain. When she joined the class action in 1994, a rheumatologist diagnosed her with atypical connective tissue disease, which he blamed on the implants.
She received an $872 payout reduced to $600 after legal bills.
The former co-ordinator of the now defunct Women’s Implant Information Network, Tanya Brennan-Hodgson, said she was “horrified” the FDA had approved silicone implants.
She said many New Zealand women were still waiting for compensation.
“Frankly, the compensation is a bit of a farce. Foreign claimants are only entitled to a pittance compared with what US women have received, and nothing can ever compensate you for the loss of your health.”

Pure Style

Yes, breast implants are sexy and classy, as hot American actress (American Pie) Tara Reid shows in this picture!

Tara Reid’s plastic surgery remained a rumour until November 4, 2004, when she was embarrassed by a wardrobe malfunction. At P. Diddy’s thirty-fifth birthday party at Cipriani’s Restaurant in New York her dress fell, exposing her left breast. Surprisingly, it took a relatively long amount of time for Reid to notice the malfunction, which led some to suspect that she was inebriated. Scar tissue was visible around Reid’s nipple, and her plastic surgery became more publicly evident…